In manual electroplating, parts are coated using the immersion process. The following base materials can be electroplated:
- aluminum
- copper
- non-ferrous metals
- stainless steel
- cast iron
The following layers can be deposited:
- Sour copper
- cyan copper
- nickel
- Electroless nickel
- silver
- tin
- anodized layer

| Beschichten/Anodisieren | Färben | |
|---|---|---|
| Länge | 800 mm | 490 mm |
| Breite | 500 mm | 390 mm |
| Höhe | 800 mm | 800 mm |
| Andere Masse | auf Anfrage |
Electroless nickel
The electroless nickel bath is an electrolyte for electroless deposition of nickel-phosphorus coatings. The deposited layers are free of lead and cadmium and are therefore RoHS-compliant. The precipitation contains 6-9% phosphorus. It is a light, semi-glossy to glossy layer. The advantages of a chemically deposited nickel layer are the even layer distribution, even on workpieces with complex geometric profiles, and the very good anti-corrosion properties.
Layer thickness recommendations according to EN ISO 4527
- Low wear and corrosion stress 5 – 10 µm
- Average wear and corrosion stress 10 – 25 µm
- High wear and corrosion stress 25 – 50 µm
- Very high wear and corrosion stress> 50 microns


Electrolytic nickel
The nickel bath meets all the requirements of a modern high-performance electrolyte. The electrolyte is optimally optimized to meet the needs of our customers in terms of corrosion protection. It is mainly used for technical interlayers.
Deposition of high-gloss, decorative layers on request .
Sour copper
The bath has good metal distribution and excellent hiding power. The copper coatings are fine-grained to moderately bright and ductile. The electrolyte is mainly used for technical intermediate layers and thick copper plating.
Deposition of high-gloss, decorative layers on request .


silver (technical)
The silver electrolyte is particularly suitable for the electrical and electronics industry.
It is characterized by its very good spreadability. The specific electrical resistance of the silver freshly separated from the silver bath is initially 1.88 µΩ x cm and, during storage, assumes the value of pure silver, 1.66 µΩ x cm. The hardness of freshly deposited silver coatings is 120 HV0.05. A value of 80 HV0.05 is reached during storage. The silver bath provides bright, shiny and haze-free layers.
High-gloss, decorative layers on request .
tin
The tin bath is an electrolyte for the deposition of shiny tin layers. Even with low current densities, eg with geometrically unfavorable workpieces, light tin layers are still deposited. The solderability of the deposited tin coating is excellent and is retained even after the aging test. The layers deposited from this electrolyte meet the requirements of the RoHS.

anodizing
Anodizing is used to create an oxidic protective layer on aluminum through anodic oxidation. In contrast to the galvanic coating process, the protective layer is not deposited. An oxide layer is formed by converting the top metal layer. After anodizing, the workpiece can be colored. In any case, it must be sealed (sealing) to ensure corrosion protection. Layer thicknesses of up to 25 µm are possible with us.
Colours:

| Schwarz | Blau | Rot | Andere Farben auf Anfrage |
|---|---|---|---|
 |  |  |
Classification of layer thickness classes according to DIN 17611 :
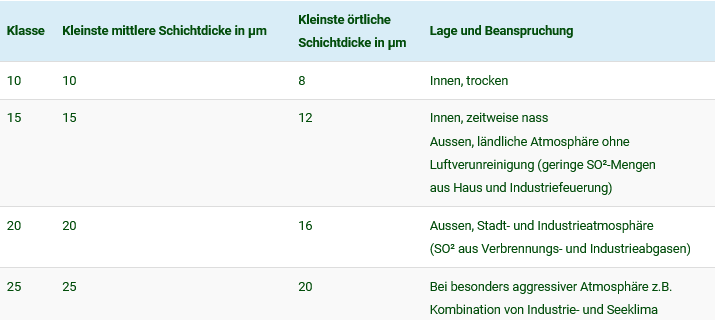
| Klasse | Kleinste mittlere Schichtdicke in µm | Kleinste örtliche Schichtdicke in µm | Lage und Beanspruchung |
|---|---|---|---|
| 10 | 10 | 8 | Innen, trocken |
| 15 | 15 | 12 | Innen, zeitweise nass Aussen, ländliche Atmosphäre ohne Luftverunreinigung (geringe SO²-Mengen aus Haus und Industriefeuerung) |
| 20 | 20 | 16 | Aussen, Stadt- und Industrieatmosphäre (SO² aus Verbrennungs- und Industrieabgasen) |
| 25 | 25 | 20 | Bei besonders aggressiver Atmosphäre z.B. Kombination von Industrie- und Seeklima |
Cyan copper
A cyanide high-performance electrolyte for the deposition of shiny, smooth, finely crystalline and ductile copper layers. This electrolyte is primarily used as a pre-copper plating electrolyte. Post-copper plating takes place in an acidic copper electrolyte. The advantage of this bath is that aluminum and steel can be coppered directly.


